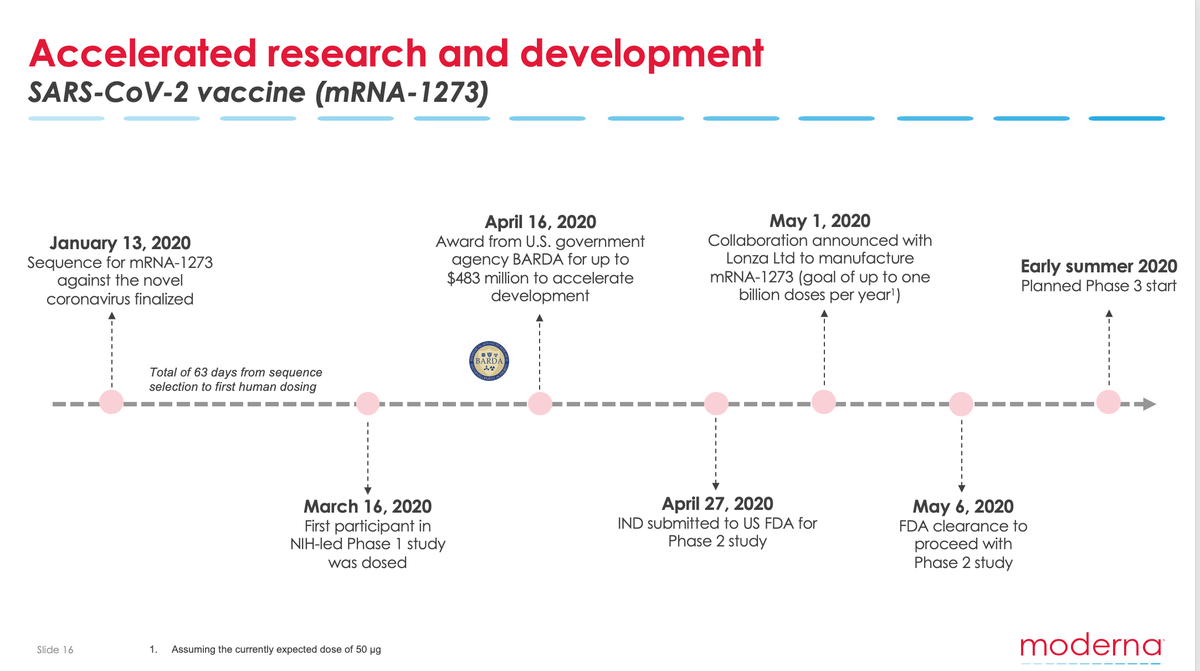
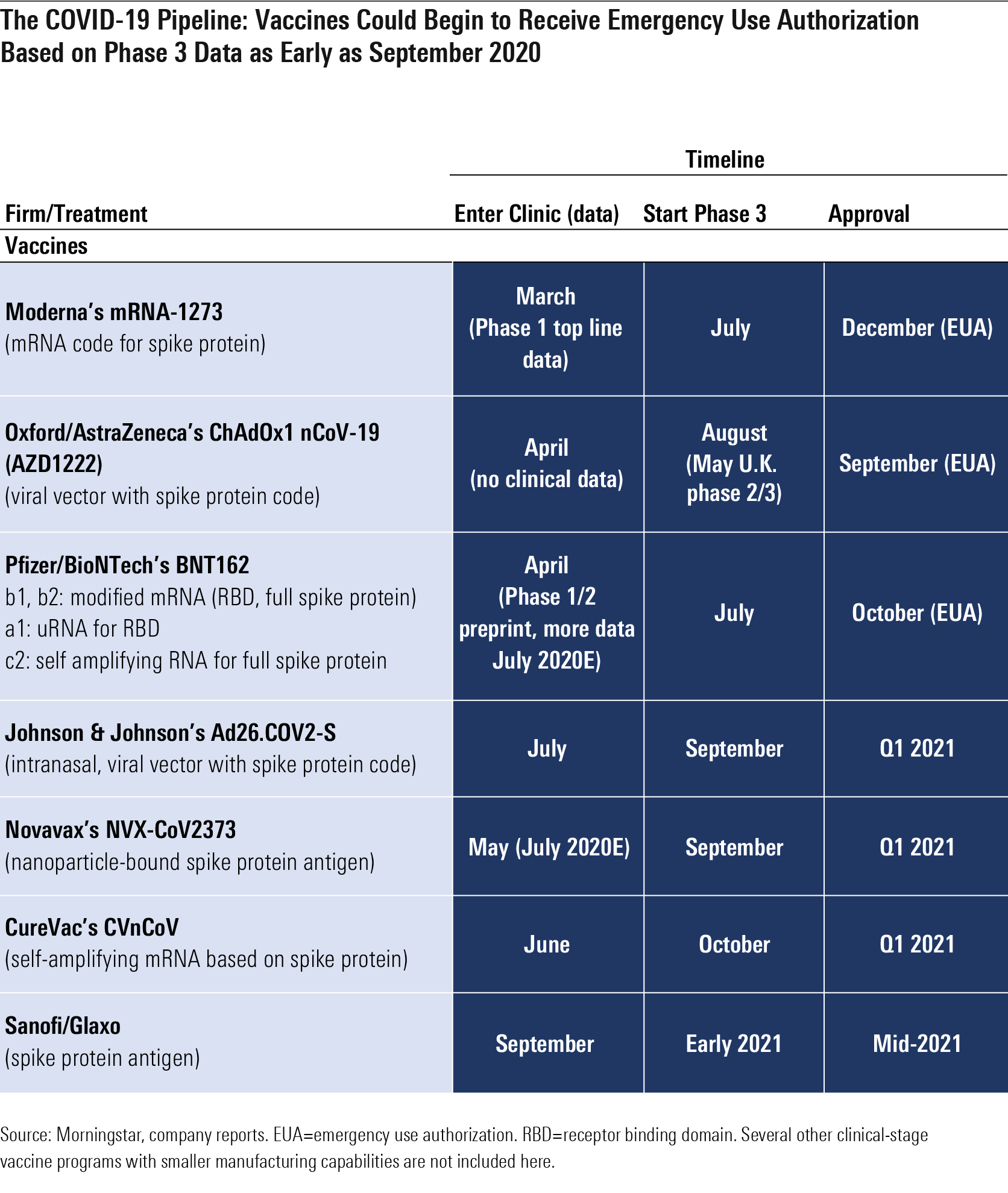
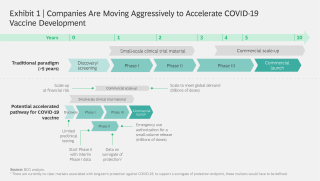
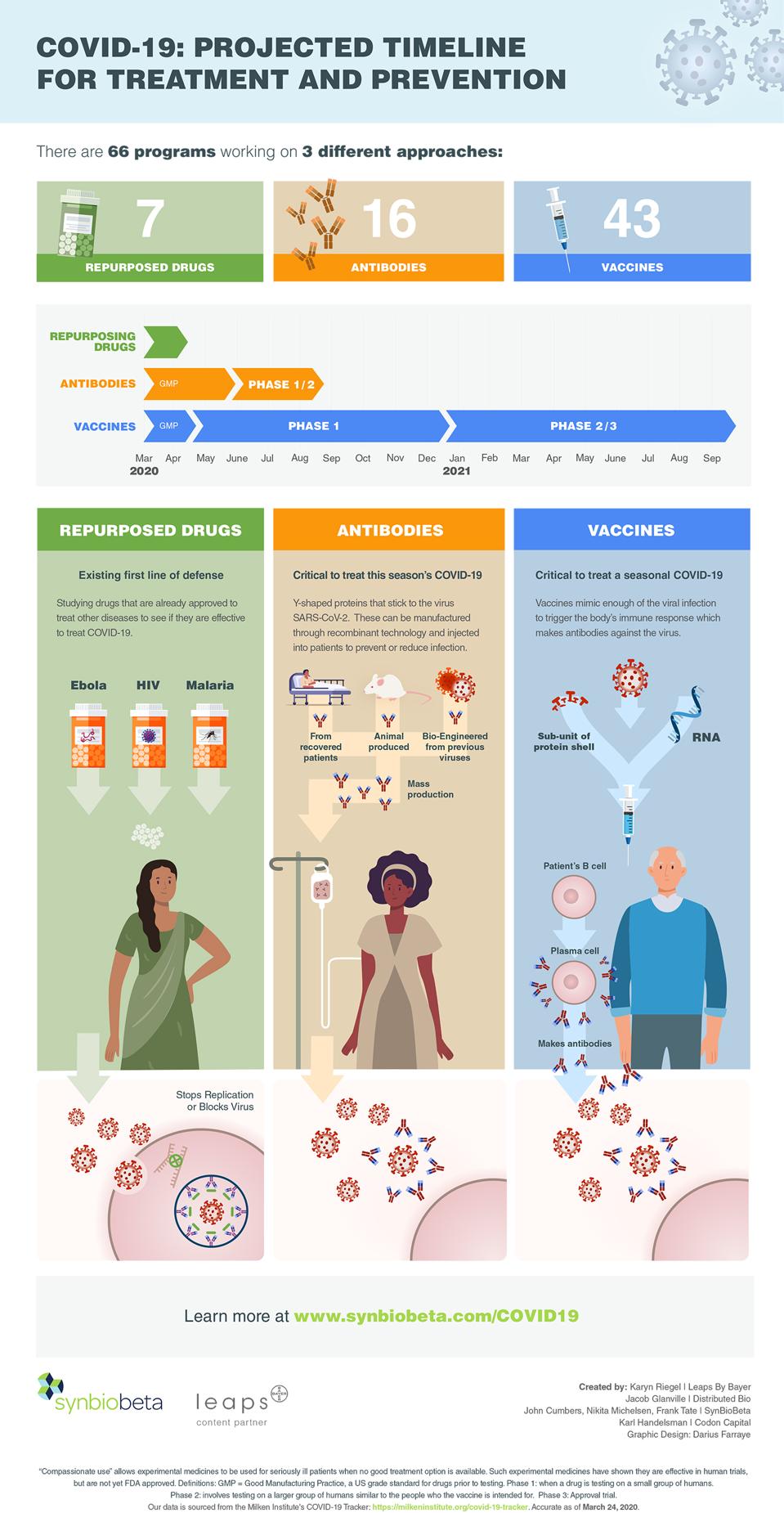
Moderna Vaccine Phase 3 Timeline
Moderna is proud to be among the many groups working to respond to this continuing global health emergency.
Moderna vaccine phase 3 timeline. A phase 3. A coronavirus vaccine being developed by moderna and the nih is the. This page summarizes key milestones in our work to advance mrna 1273 our vaccine candidate against the novel coronavirus. The vaccine known as mrna 1273 was co developed by the cambridge massachusetts based biotechnology company moderna inc and the national institute of allergy and infectious diseases niaid part of the national institutes of health.
The three companies are among nine drugmakers that have. Phase 3 moderna develops vaccines based on messenger rna mrna to produce viral proteins in the body. Coronavirus vaccine to reach the final phase 3. Phase three is when human trials are conducted and could take between 5 and 9 years as scientists figure out if the vaccine is safe activates an immune response and protects against the disease.
Learn more about mrna 1273 moderna s covid 19 vaccine candidate. A vaccine against this pandemic virus. In january they began developing a vaccine for the. These trials will determine if.
O n june 11 biotech company moderna announced it had finalized plans for phase 3 testing of its covid 19 vaccine candidate. Timeline accounts for the few weeks it will. Moderna s ceo gave a detailed timeline for when we ll know if its coronavirus vaccine works and cautions that you might not get a shot until the spring what phase 3 means. They have yet to bring one to the market.
Mrna announced that it has started phase 3 trials of its covid 19 vaccine candidate mrna 1273 in collaboration with two u s. The late stage trial will include 30 000 participants and is expected.