Phase 3 Moderna Vaccine
Find out why this vaccine technology is promising but not without its skeptics.
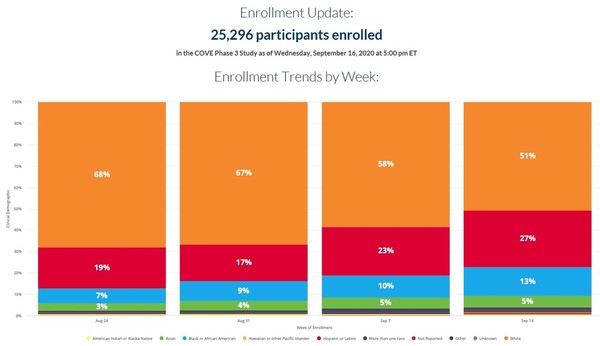
Phase 3 moderna vaccine. Moderna is looking to start phase 3 trials for the vaccine this month and has indicated that it could have data from the study by thanksgiving. The biotech firm s leading candidate has just entered phase three clinical trials in the u s. Updated 7 6 2020 the recent moderna selloff. Moderna said preliminary phase three trial data shows its coronavirus vaccine is more than 94 effective in preventing covid 19.
The vaccine known as mrna 1273 was co developed by the cambridge massachusetts based biotechnology company moderna inc and the national institute of allergy and infectious diseases niaid part of the national institutes of health. Moderna have announced that their vaccine candidate against covid 19 has demonstrated 94 5 efficacy in the first interim analysis from the phase 3 vaccine trials. The same dsmb also oversees the additional ows supported phase 3 clinical trials evaluating covid 19 vaccine candidates. Prof adrian hill professor of human genetics director of the jenner institute co director oxford martin programme on vaccines and fellow of magdalen college said.
Moderna said that phase 3 stage analysis suggested its coronavirus vaccine could prevent covid 19. The news comes a week after pfizer and german drug maker biontech announced a vaccine of their own. In january they began developing a vaccine for the. The company s ceo stephane bancel called it a game changer.
Moderna announces initial phase 3 data showing its covid 19 vaccine is up to 94 5 effective moderna s vaccine can be stored in a standard refrigerator unlike pfizer s. As part of the arrangement under ows representatives from niaid barda and moderna are part of the oversight group that receives recommendations from the trial s independent dsmb. They have yet to bring one to the market.