Moderna Vaccine Volunteer Phase 3
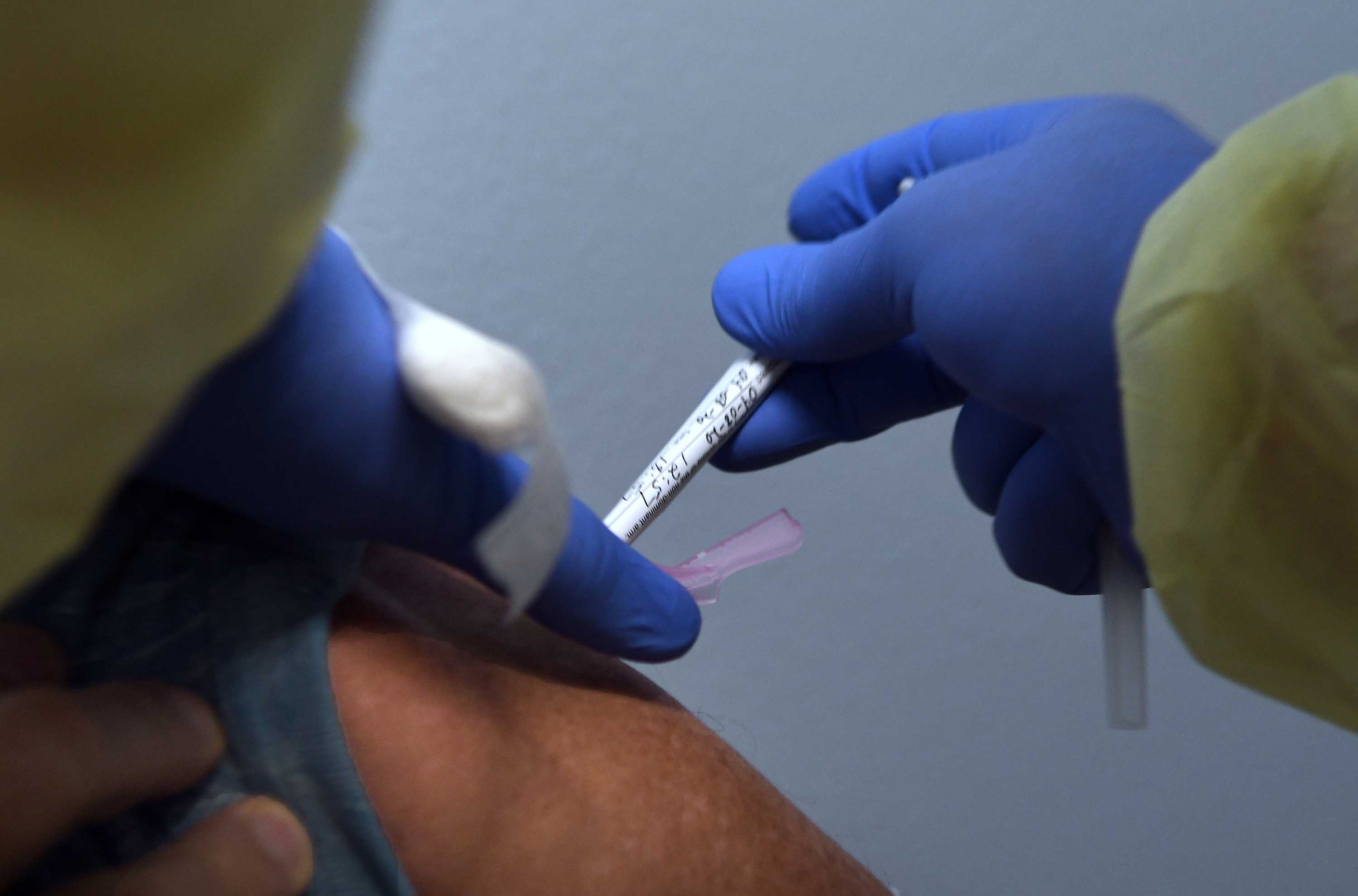
The department of veterans affairs is recruiting 8 000 volunteers for the phase 3 clinical trials of at least four covid 19 vaccine candidates at 20 federal medical facilities across the u s.
Moderna vaccine volunteer phase 3. Moderna covid vaccine begins phase 3 03 30. The late stage trial will include 30 000 participants and is expected. O n june 11 biotech company moderna announced it had finalized plans for phase 3 testing of its covid 19 vaccine candidate. A phase 3 clinical trial designed to evaluate if an investigational vaccine can prevent symptomatic coronavirus disease 2019 covid 19 in adults has begun.
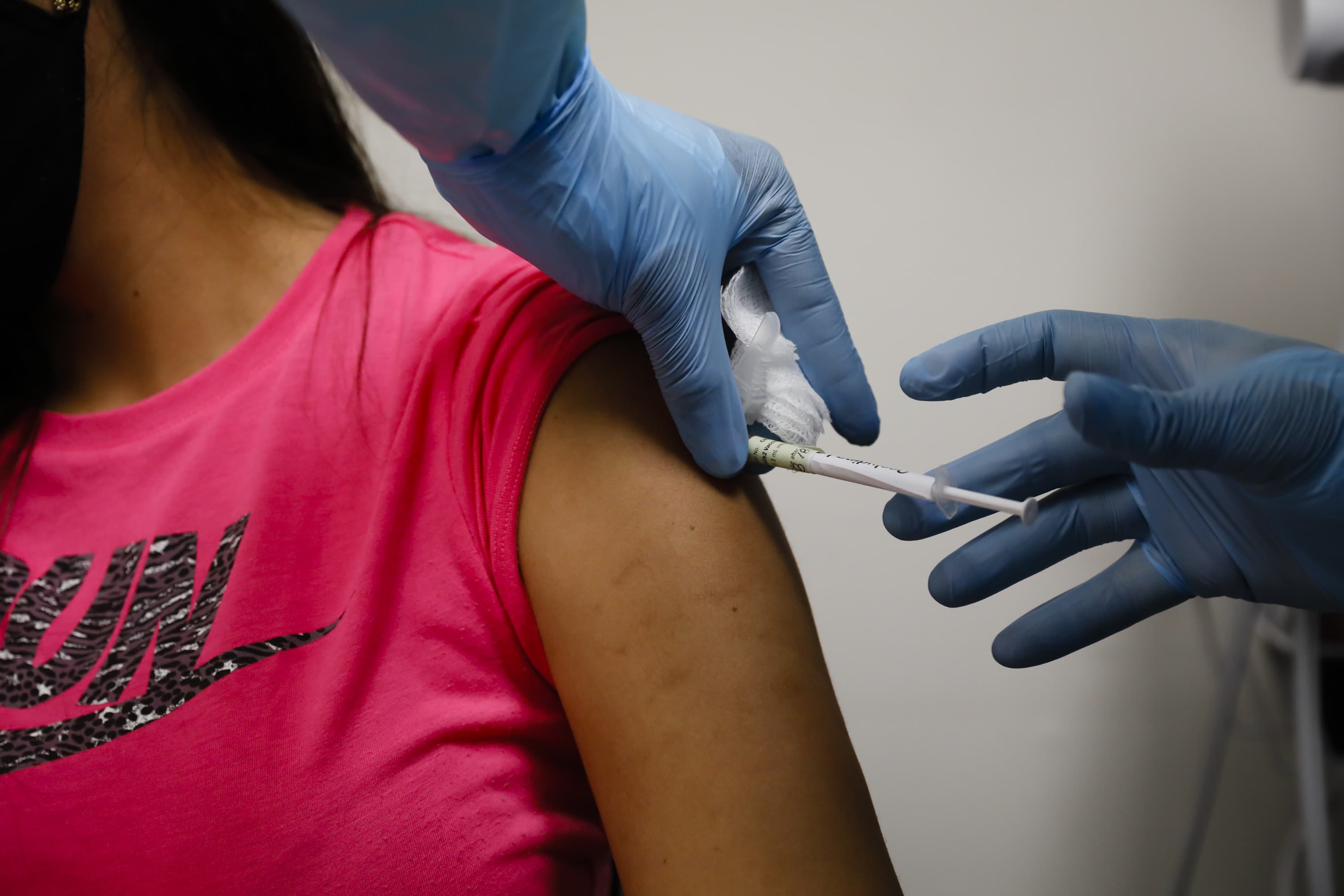
Half will receive two shots of the vaccine 28 days apart while the other half will receive two. In the race for the vaccine moderna announced it s on track to enroll 30 000 people for its phase three trial by the end of september. Moderna is committed to advancing the clinical development of mrna 1273 as safely and quickly as possible to demonstrate our vaccine s ability to significantly reduce the risk of covid 19 disease vaccine candidate mrna 127 is currently in a phase ii clinical trial which will enrol 600 healthy participants aged 18 and above. The phase 3 trial.
The phase three clinical trial of moderna s vaccine candidate which began last month on july 27 is expecting to recruit around 30 000 adult volunteers. The world s biggest covid 19 vaccine study is now underway with the first of 30 000 planned volunteers helping to test shots created by the u s. The vaccine known as mrna 1273 was co developed by the cambridge massachusetts based biotechnology company moderna inc and the national institute of allergy and infectious diseases niaid part of the national institutes of health. In the moderna trial 30 000 volunteers will be followed for.
The phase 3 vaccine efficacy trial known as cove was begun under ows a multi agency collaboration led by hhs and the department of defense that aims to accelerate the development manufacturing and distribution of medical countermeasures for covid 19.