Moderna Vaccine Trial Questioned

Major averages fell to session lows in the wake of a report from stat news that questioned the validity of the results of moderna s vaccine trial which the company had announced on monday us time.
Moderna vaccine trial questioned. Astrazeneca and oxford defend covid vaccine trials after questions raised in the u s. According to meridian clinical research which is helping to organize the phase 3 clinical trial for the moderna vaccine volunteers must be at least 18 years of age with no known history of. The same dsmb also oversees the additional ows supported phase 3 clinical trials evaluating covid 19 vaccine candidates. Moderna has yet to mention when the vaccine will be available to.
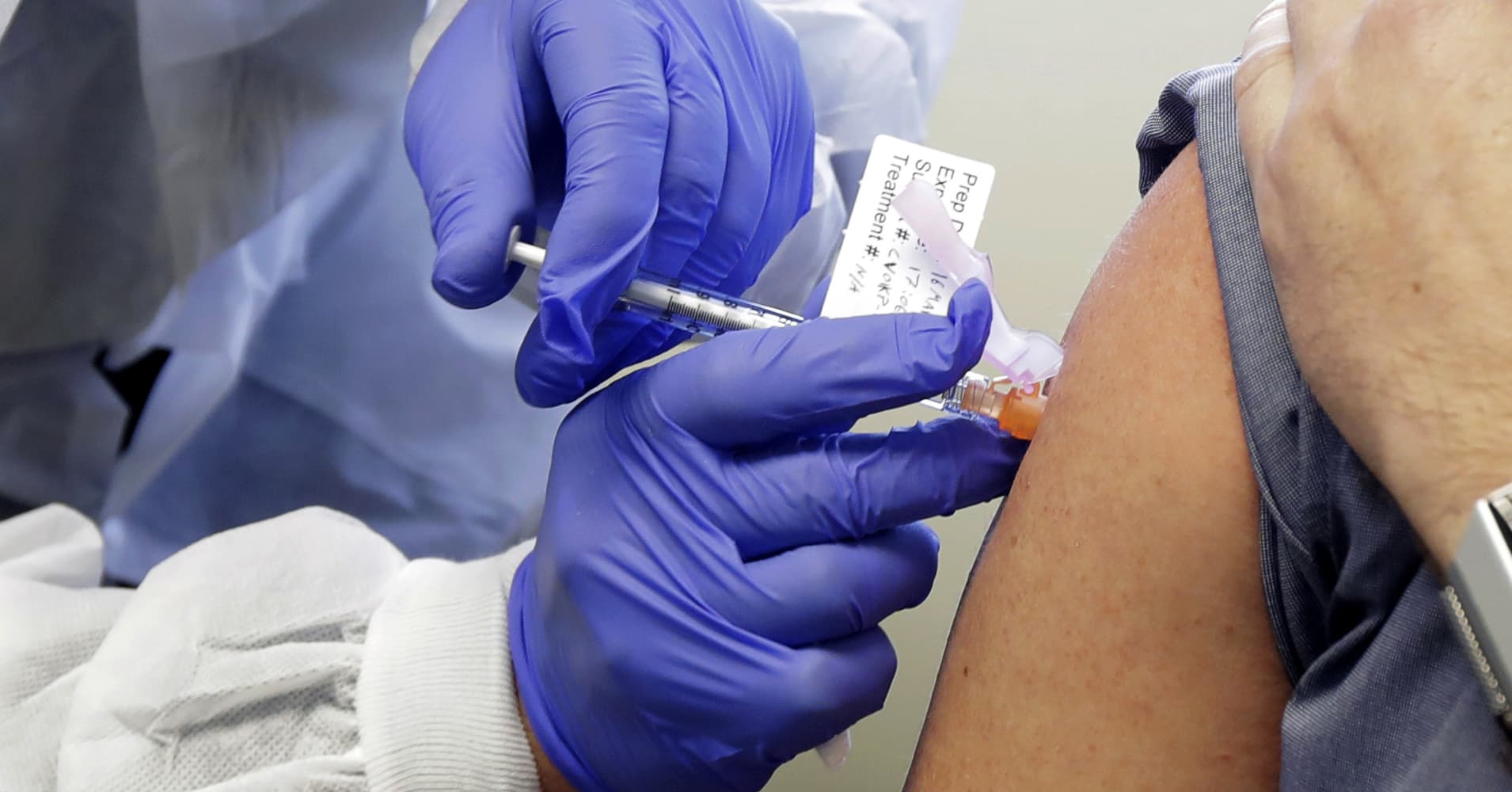
Published thu nov 26 2020 7 57 am est updated thu nov 26 2020 11 48 am est natasha turak natashaturak. A nurse prepares a shot as the world s biggest study of a possible covid 19 vaccine developed by the national institutes of health and moderna gets underway on jul 27 2020 in binghamton new york. Moderna s clinical trial began recruiting participants in late july so there is only evidence thus far that people who received the vaccine have protection for a few months. The results come hot on the heels of similar results from pfizer and add to growing.