Moderna Vaccine Production
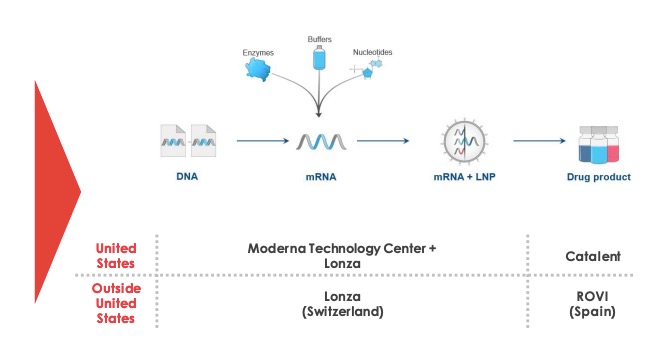
Lonza group ag made its first commercial batch of the main ingredient in moderna inc s covid 19 vaccine candidate in the us last week and plans to start european production by the end of the.
Moderna vaccine production. Baehny identified more challenges to vaccine production that the company has had to confront since embarking on its partnership with moderna. Moderna s covid vaccine found 94 5 effective in early analysis. The lonza plant at visp deep in the swiss alps is a hive of activity. Most of the production of the drug substance for moderna s vaccine is running via lonza a swiss company with a long history of.
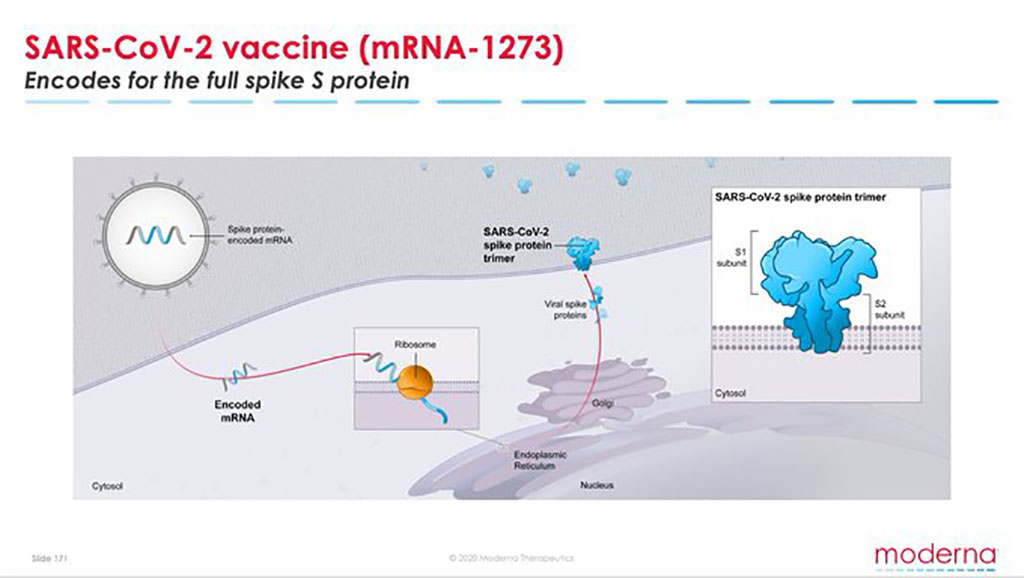
Biotech moderna nasdaq mrna announced an agreement with swiss drugmaker lonza group to manufacture up to a billion doses per year of mrna 1273 moderna s vaccine candidate for the novel coronavirus. Moderna is pushing forward with manufacturing. Moderna itself has one production line with capacity for 100 million doses a year moderna has said it will be able to deliver 20 million doses of vaccine by the end of this year enough to. Mrna 1273 is an mrna vaccine candidate against covid 19 encoding for a prefusion stabilized form of the spike s protein which was selected by moderna in collaboration with investigators from vaccine research center vrc at the national institute of allergy and infectious diseases niaid a part of the national institutes of.
There are a few issues the first is speed. Workers are racing to set up production lines to be able to start making a covid 19 vaccine for us firm moderna later this year. About mrna 1273 moderna s vaccine candidate against covid 19. Last week and plans to start european production by the end of the month chairman albert baehny said the company is ramping up and fine tuning its production lines a process that may stretch into next year we have access to the raw materials.