Moderna Vaccine Manufacturing

Ctlt the leading global provider of advanced delivery technologies development and manufacturing solutions for.
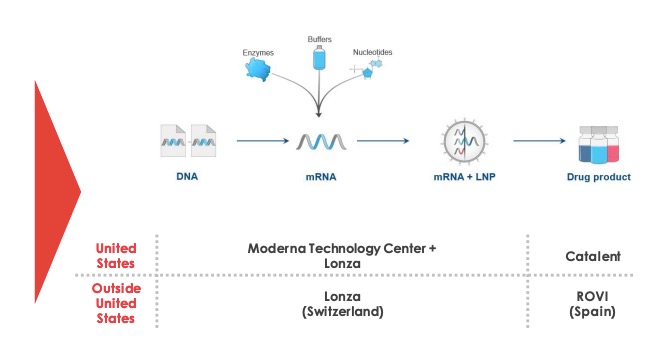
Moderna vaccine manufacturing. However the vaccine candidate. The rollout will be quick because moderna has already begun manufacturing of the vaccine and has said that it plans to have 20 million doses by the end of 2020. Biotech moderna nasdaq mrna announced an agreement with swiss drugmaker lonza group to manufacture up to a billion doses per year of mrna 1273 moderna s vaccine candidate for the novel coronavirus. Mrna a clinical stage biotechnology company pioneering messenger rna mrna therapeutics and vaccines to create a new generation of transformative medicines for patients and catalent inc.
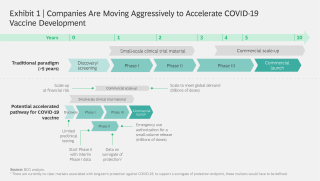
Moderna s vaccine reached phase i human trials on march 16 only 63 days after the company began developing it. Moderna s covid vaccine found 94 5 effective in early analysis. Moderna is pushing forward with manufacturing the potential vaccine called mrna 1273 in hopes that it proves safe for humans and effective against the coronavirus. Moderna and its partners in the federal government are just now gearing up for a late stage covid 19 vaccine trial set to launch later this month.
And somerset n j june 25 2020 moderna inc. Most of the production of the drug substance for moderna s vaccine is running via lonza a swiss company with a long history of. But manufacturing went faster than expected and by feb. Baehny identified more challenges to vaccine production that the company has had to confront since embarking on its partnership with moderna.
But at the same time the mrna biotech is. Moderna has already started to prepare for rapid acceleration of its manufacturing capabilities that could allow for the future manufacture of millions of doses should mrna 1273 prove to be safe and of expected benefit.