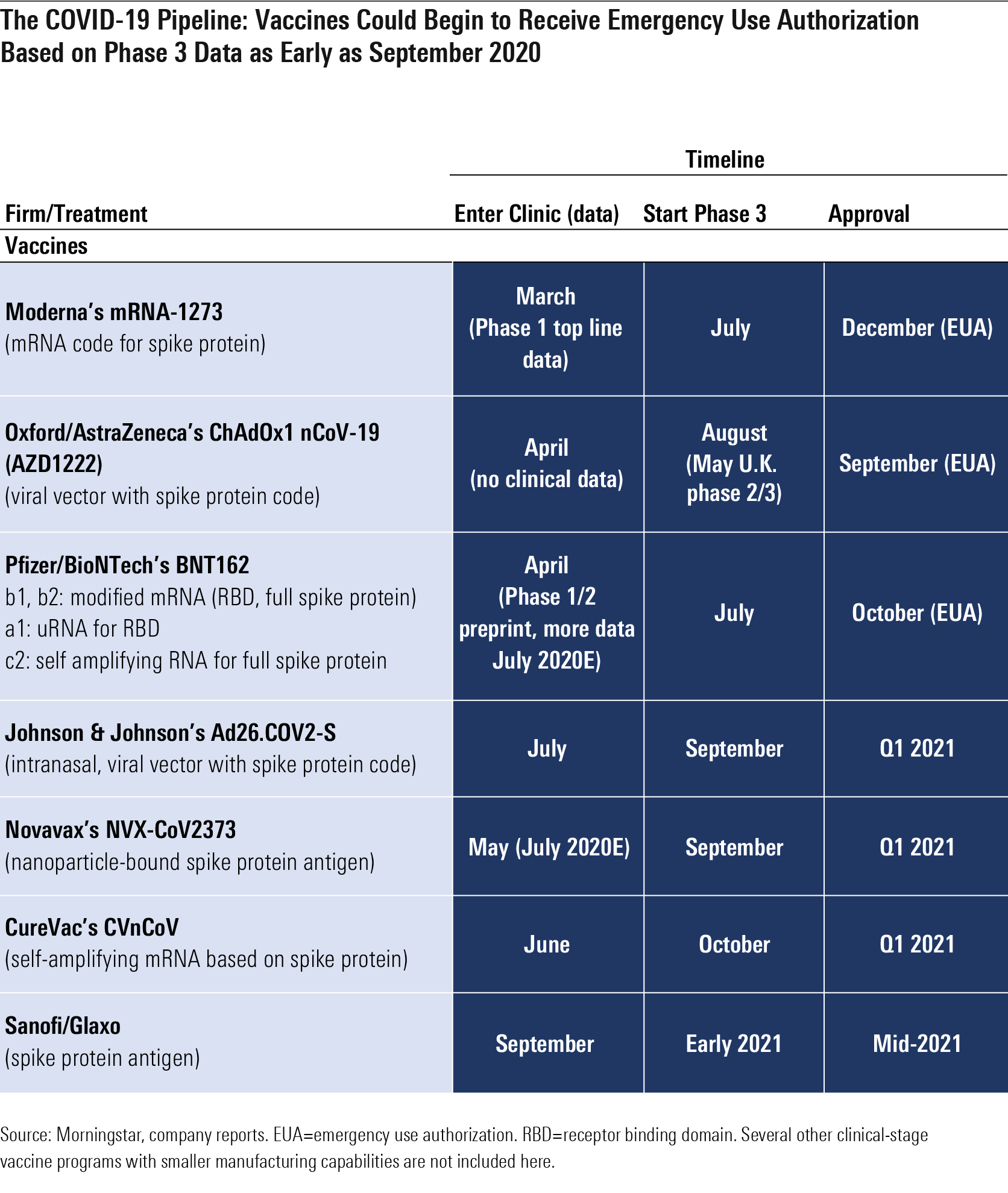
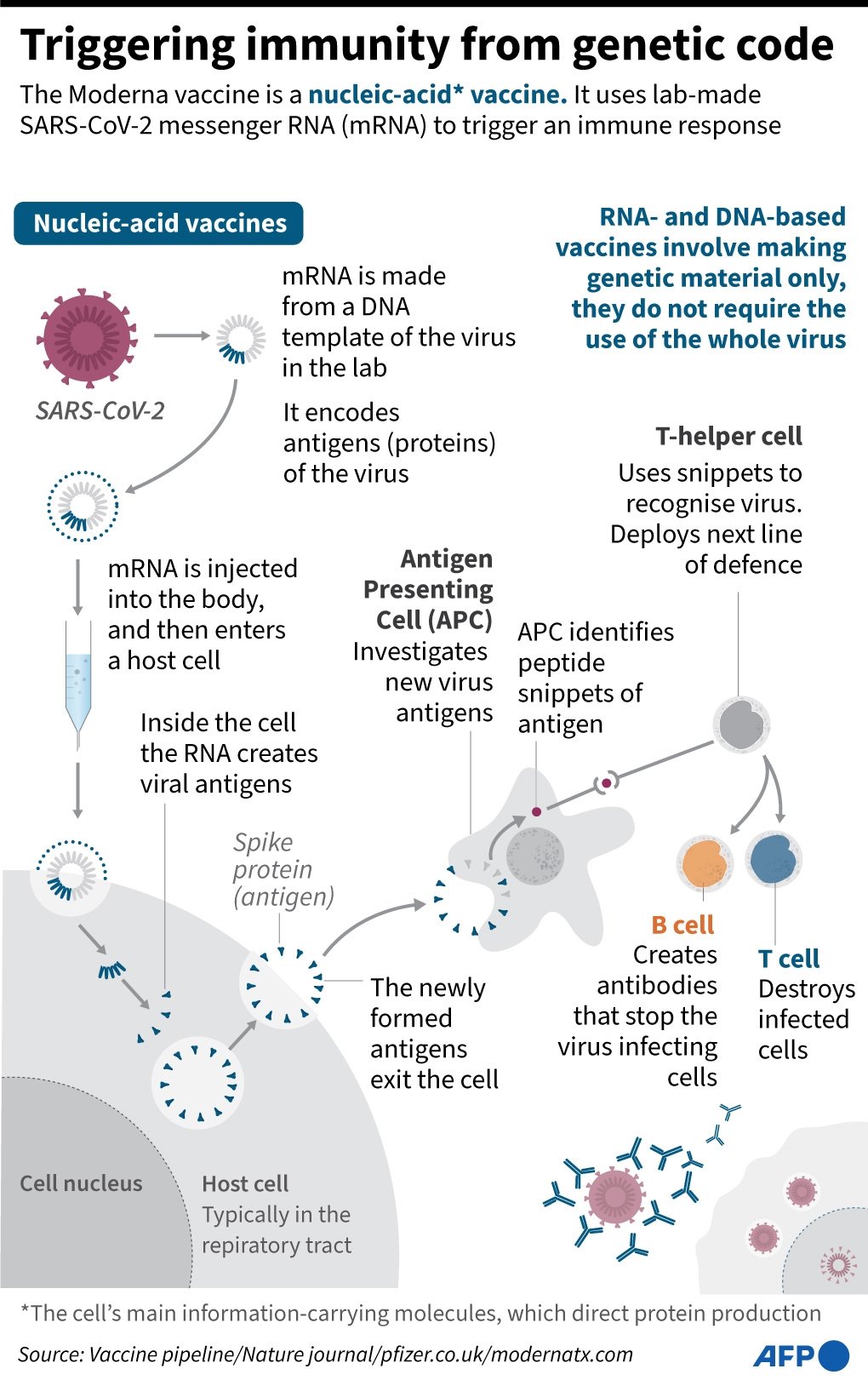
Moderna Vaccine Journal

Mrna a biotechnology company pioneering messenger rna mrna therapeutics and vaccines to create a new generation of transformative medicines for patients today announced the publication of the second interim analysis of the open label phase 1 study of mrna 1273 its vaccine candidate against covid 19 in the new.
Moderna vaccine journal. Moderna said the analysis of the phase three cove study of the vaccine candidate called mrna 1273 involving 30 000 participants included 196 cases of covid 19 of which 30 cases were severe. The fda is set to review the vaccine for emergency. Moderna inc s experimental vaccine is 94 5 effective in preventing covid 19 based on interim data from a late stage trial the company said on monday becoming the second u s. 29 2020 moderna inc nasdaq.
Moderna s vaccine appears to be 94 1 successful in its phase 3 trial at preventing covid 19 and 100 successful at preventing severe cases. Mrna vaccine against covid 19 in older adults the moderna mrna 1273 vaccine which elicited antibodies and t cells specific for the covid 19 virus in adults 55 years of age or younger elicited sim. Health moderna and pfizer are reinventing vaccines starting with covid the two leading vaccines including one developed by biontech are based on a new gene based technology that could help. In this july 27 2020 file photo nurse kathe olmstead prepares a shot that is part of a possible covid 19 vaccine developed by the national institutes of health and moderna inc in binghamton.