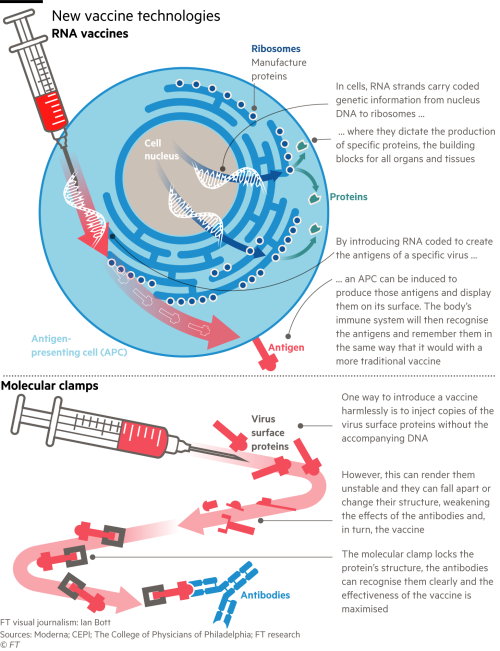
Moderna Vaccine Failure

A sign marks the headquarters of moderna therapeutics which is developing a vaccine against the coronavirus disease covid 19 in cambridge massachusetts u s may 18 2020.
Moderna vaccine failure. The company said in a press release that the first interim analysis from the phase 3 cove randomized controlled trial found a total of 95 covid 19 cases 90 of which were in the. The company said it would immediately seek. The breakthrough vaccine platform they use. Moderna inc s mrna o experimental vaccine is 94 5 effective in preventing covid 19 based on interim data from a late stage trial the company said on monday becoming the second u s.
H eavy hearts soared monday with news that moderna s covid 19 vaccine candidate the frontrunner in the american market seemed to be generating an immune response in phase 1 trial. Earlier this week moderna said that its. The new technology behind pfizer s and moderna s coronavirus vaccines could be used to prevent everything from heart disease to cancer experts say. Announced this morning that early data from a late stage clinical trial indicate its vaccine candidate showed 94 5 efficacy against the coronavirus.
Said monday that its. The biotech company moderna inc. The news comes a week after pfizer and german drug maker biontech announced a vaccine of their own. The company s ceo stephane bancel called it a game changer.
Moderna stock is up by about 270 year to date as it emerged a front runner in the race to develop a vaccine for the highly contagious novel coronavirus. A second covid 19 vaccine now also appears highly effective in preventing illness following exposure to the virus that causes the disease. Moderna said monday that its covid 19 vaccine continued to deliver strong efficacy results showing 94 efficacy in the main analysis of its key study.