Moderna Vaccine Enrollment

We are indebted to all of the participants in the study said stéphane bancel chief executive officer of moderna.
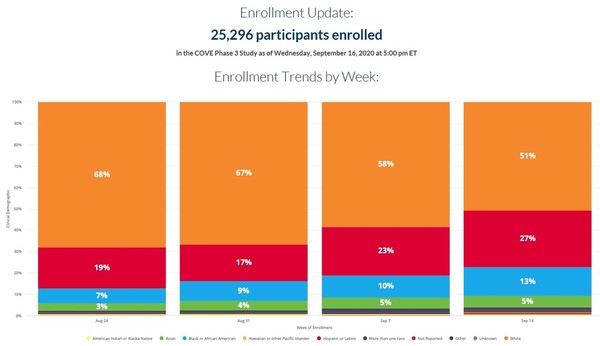
Moderna vaccine enrollment. Moderna one of the developers in the lead for a vaccine to prevent covid 19 is slowing enrollment slightly in its large clinical trial to ensure it has sufficient representation of minorities. Moderna s coronavirus vaccine trial enrollment nears completion the company s chief executive officer indicated that moderna will pursue an emergency use authorization for high risk groups if. Moderna s coronavirus vaccine trial is fully enrolled with people of color making up more than one third of the 30 000 participants the drugmaker announced thursday as government scientists. Moderna inc said on thursday it had completed the enrollment of 30 000 participants in a late stage study testing its experimental coronavirus vaccine with over a third of the participants from.
28 the moderna cove study had enrolled more than 17 000 people and has testing sites all over the country. Mrna which is one of the three companies outside of china to have moved its coronavirus vaccine candidates into late stage trials is close to completing targeted enrollment. Moderna s vaccine candidate is in a large scale phase 3 trial dubbed cove with a target enrollment of 30 000 participants about 28 043. Learn more about mrna 1273 moderna s covid 19 vaccine candidate.
Learn about mrna 1273 moderna s potential vaccine against covid 19 study participation overview while enrolled in the cove study participants will need to attend study visits and comply with study requirements which include completing diary entries and immediately contacting the study physician if they are experiencing symptoms of covid 19. Moderna s slower enrollment rate. Moderna is proud to be among the many groups working to respond to this continuing global health emergency. Moderna vaccine trial sign up.