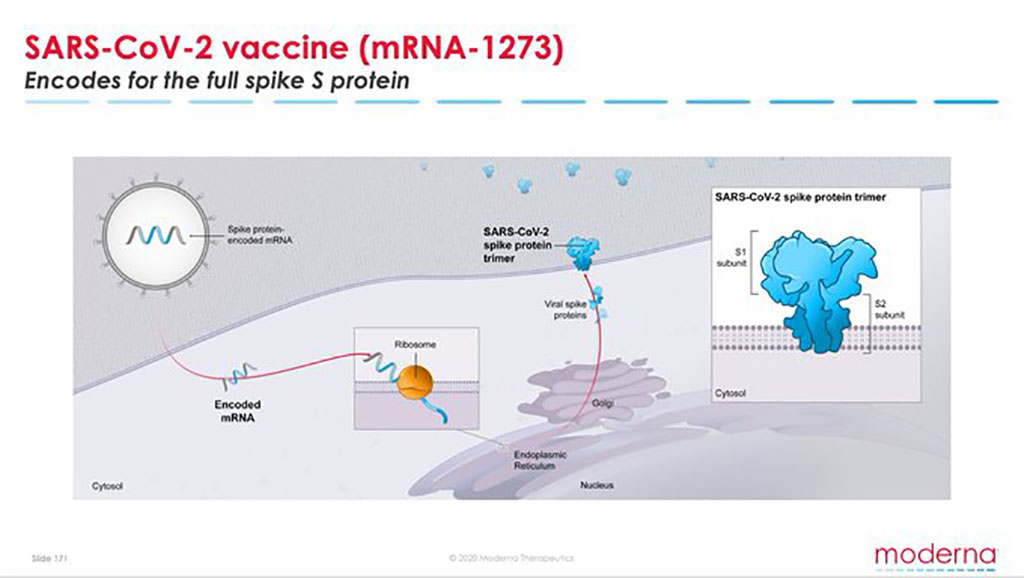
Moderna Vaccine Data

Moderna s covid 19 vaccine is strongly effective early look at data show recommended osha s absurd reinterpretation of a regulation regarding workers and.
Moderna vaccine data. Washington moderna s experimental vaccine was 94 5 per cent effective in preventing covid 19 based on interim data from a late stage trial the company said yesterday as it became the second. The fda requires at least 2 months of safety data following the second dose. Moderna s covid 19 vaccine is 94 5 effective in preventing the disease preliminary data from large scale phase three trials shows the company announced in a press release on monday. Like the pfizer biontech vaccine the moderna vaccine will also require cold storage which experts have noted could pose problems for distribution.
Preliminary vaccine data from pfizer and moderna are encouraging but there s more steps to travel before the vaccines are available. Moderna announces initial phase 3 data showing its covid 19 vaccine is up to 94 5 effective moderna s vaccine can be stored in a standard refrigerator unlike pfizer s. The moderna vaccine is 94 5 effective against coronavirus according to early data released monday by the company making it the second vaccine in the united states to have a stunningly high. The results come hot on the heels of similar results from pfizer and add to growing.
Clinical data for moderna s covid 19 vaccine showed it was nearly 95 effective in preventing disease according to an interim analysis described in a company release monday. Moderna said it expects at least 53 people in the study contracted the virus the minimum number required for an independent data safety monitoring board to review and measure how well the vaccine.






.1589833729140.png?)